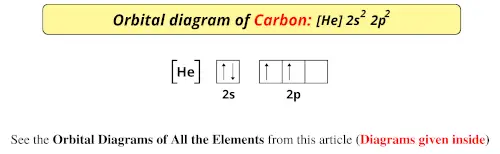
It determines many physical and chemical properties of that atom. The helium atom contains two protons and two electrons. The specific arrangement of electrons in atomic orbitals is called the electron configuration of the atom. To do that we need to find the number of. Lets find the ground state electron configuration of Carbon In this example video we are asked about carbon ground state electron configuration A single c. 
Construct an orbital diagram to show the electron. When the hybridization state of carbon atom changes from s p 3 to s p 2 to sp. In writing the electron configuration for carbon the first two electrons will go in the 1s orbital. an orbital diagram or an electron configuration 11. The electron configuration and the orbital diagram are: Following hydrogen. The electron configuration and the orbital diagram are:įollowing hydrogen is the noble gas helium, which has an atomic number of 2. To write the orbital diagram for the Carbon atom (C) first we need to write the electron configuration for just C. If we added one more electron to borons orbital diagram we will get carbons orbital diagram. Atomic number of carbon, Z6 Use the order of fill diagram to draw an orbital filling diagram with a total of six electrons. Step 1: List the known quantities and plan the problem. An orbital box diagram can be written as well. Draw the orbital filling diagram for carbon and write its electron configuration. electronic configuration of Carbon ( 6 C) is: 1s2 2s2 2p2. The electron configuration for carbon is 1s 2 2s 2 2p 2. Electron configurations of atoms follow a standard. To write electron configurations in orbital notation, find the element on the periodic table. It is worth noting that the arrangement of electrons in different 2 p orbitals, necessitated by Hund’s rule, produces a configuration of lower energy.\) value is usually filled first. The electron configuration of an element describes how electrons are distributed in its atomic orbitals. In such a case it is assumed that the reader knows that the two 2 p electrons are not spin paired.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
